Fog and dew, on the other hand, do not come within the category of precipitation. Precipitation includes both heavy and mild rain, as well as snow, drizzle, and hail. The cooling of air molecules and the addition of water vapor are the two processes that cause the air to become saturated. 
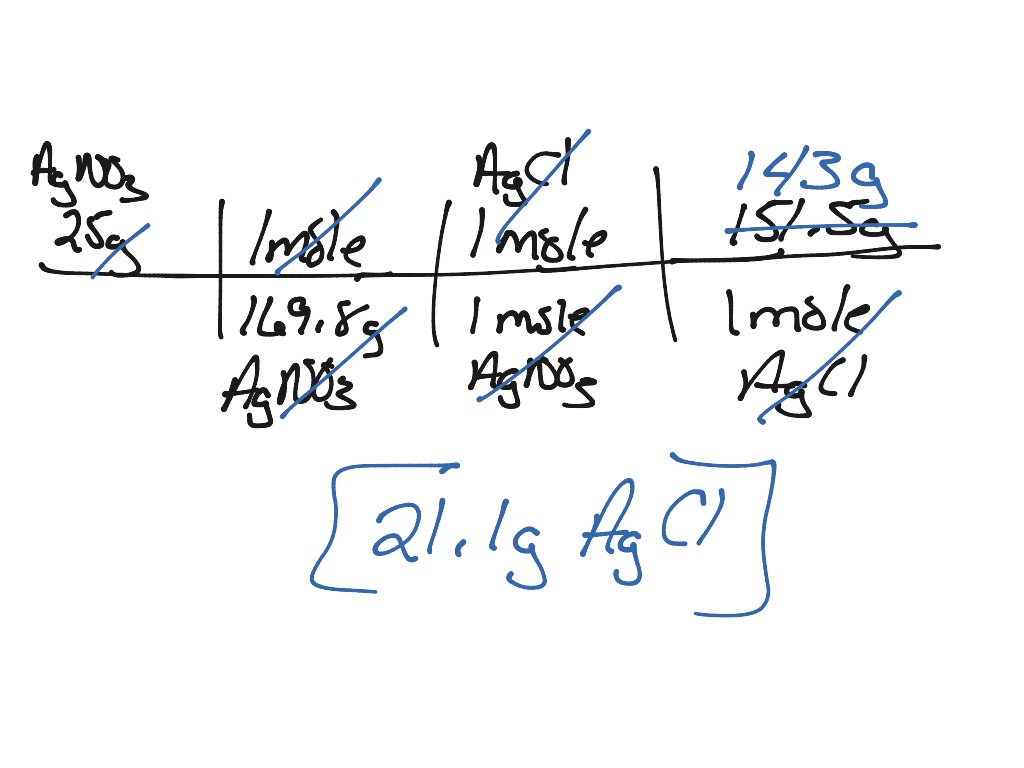
Precipitation, for example, occurs when a portion of the atmosphere becomes saturated with water vapor, which then condenses and precipitates when the temperature is just perfect. It's a chemical reaction in which two ionic material solutions are combined to generate a solid ionic substance (a precipitate). Kinetic effects (like speed of crystallization and presence of mixing) play a role.Precipitation is defined as any liquid or frozen water that exist in the atmosphere and falls to the Earth's surface. This is the case when diffusion in the interior is possible (like in the liquids) or when the initial small crystals are allowed to recrystallize. When the Berthelot-Nernst law applies, then the concentration of the tracer in the interior of the crystal is uniform (and the crystals are said to be homogeneous). When this assumption is fulfilled, then the content of the tracer in the crystal is non-uniform (the crystals are said to be heterogeneous). The derivation of the Doerner-Hoskins law assumes that there in no mass exchange between the interior of the precipitating crystals and the solution. Ln a a − x = λ ln b b − y Ī and b are the initial concentrations of the tracer and carrier, respectively a − x and b − y are the concentrations of tracer and carrier after separation x and y are the amounts of the tracer and carrier on the precipitate D and λ are the distribution coefficients.įor D and λ greater than 1, the precipitate is enriched in the tracer.ĭepending on the co-precipitation system and conditions either λ or D may be constant. There are two models describing of the distribution of the tracer compound between the two phases (the precipitate and the solution): 
Distribution between precipitate and solution Ĭoprecipitation is also used as a method of magnetic nanoparticle synthesis. An occlusion occurs when an adsorbed impurity gets physically trapped inside the crystal as it grows.īesides its applications in chemical analysis and in radiochemistry, coprecipitation is also important to many environmental issues related to water resources, including acid mine drainage, radionuclide migration around waste repositories, toxic heavy metal transport at industrial and defense sites, metal concentrations in aquatic systems, and wastewater treatment technology. An adsorbate is an impurity that is weakly, or strongly, bound ( adsorbed) to the surface of the precipitate. An inclusion (incorporation in the crystal lattice) occurs when the impurity occupies a lattice site in the crystal structure of the carrier, resulting in a crystallographic defect this can happen when the ionic radius and charge of the impurity are similar to those of the carrier. There are three main mechanisms of coprecipitation: inclusion, occlusion, and adsorption. Otto Hahn is credited for promoting the use of coprecipitation in radiochemistry. An example is the separation of francium from other radioactive elements by coprecipitating it with caesium salts such as caesium perchlorate. Since the trace element is too dilute (sometimes less than a part per trillion) to precipitate by conventional means, it is typically coprecipitated with a carrier, a substance that has a similar crystalline structure that can incorporate the desired element. On the other hand, in the analysis of trace elements, as is often the case in radiochemistry, coprecipitation is often the only way of separating an element. Typical co-precipitation method for micro and nano particle synthesis 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
